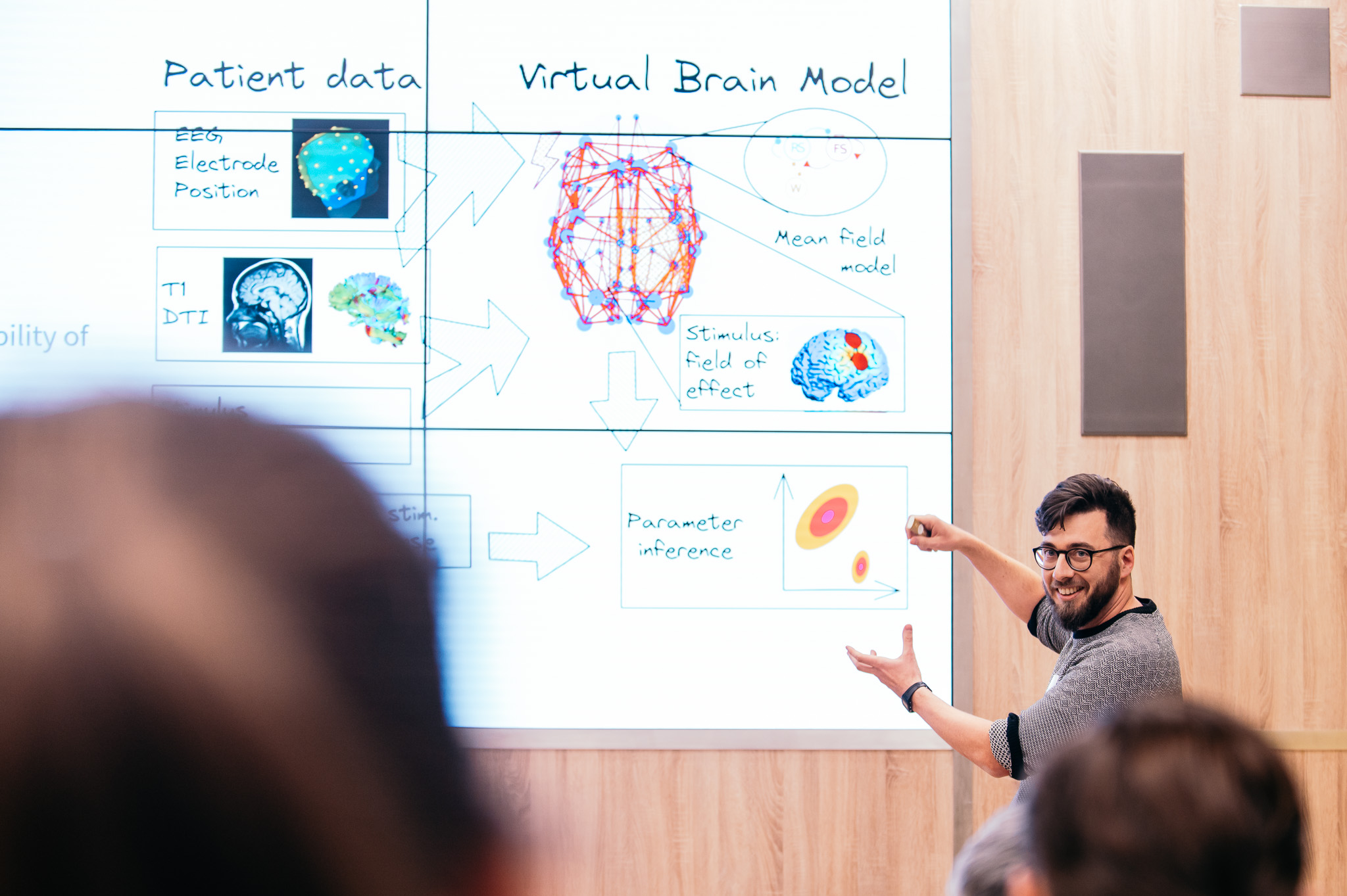
As a challenging 2021 fades, new challenges lie ahead

Like many others, MultiplexDX also had a challenging 2021. Despite all the hardship and hurdles that the pandemic continues to bring, it has also taught us to persevere and opened doors for new opportunities. We developed several new COVID-19 tests, solved scientific challenges, published in prestigious journals, and welcomed new colleagues into the MultiplexDX family, effectively doubling our team in a year. Here are some highlights from 2021 that pleased us most…
LAMP TESTS = SAFER EVENTS
Last year, we expanded our diagnostic portfolio with a new LAMP test, which has the processing complexity and speed closer to antigen tests while retaining sensitivity and accuracy that approaches RT-qPCR tests.
LAMP tests are suitable for testing at conferences, sporting events, weddings, and other events where point-of-care testing is needed.
In 2021 we provided our LAMP tests and our mobile testing solution for the international GLOBSEC Forum conference, Pohoda music festival, ICF Canoe Slalom World Championship, the Olympic Hockey Qualifiers, and more. If you have two minutes, watch our video from these events.
MULTIPLEXDX TESTS HELP WORLDWIDE
Every scientist is pleased when their hard work gets recognition and can help others. We are particularly pleased by the success of our tests in fighting the pandemic worldwide. Our tests have been registered, validated, and certified in 26 countries, including the prestigious RIVM National Reference Laboratory in the Netherlands.
SCIENTIFIC ACHIEVEMENTS
Publications from our team members in renowned scientific journals provide evidence that our research also brings new insights and knowledge. These range from our role in developing a test to identify and monitor the prevalence of the SARS-CoV-2 variants in Slovakia published in Scientific Reports and Virus Genes, to our efforts to introduce novel technologies such as fluorescently labeled sequencing adaptors as described in Nucleic Acids Research. In addition, another publication on the complete development and validation of our RT-qPCR tests is under peer-review in the prestigious international journal Microbial Biotechnology.
WE SHARE OUR SUCCESS
We donated 250,000 tests, 100,000 of which went abroad via humanitarian aid (Kenya, Ukraine, Serbia, Vatican, Montenegro, Moldova). At the end of the year, we handed over a check for EUR 339 000 to our research partners at the Biomedical Center of the Slovak Academy of Sciences.
WHAT'S AHEAD OF US? RESEARCH!
Our work on Multiplex8+, a new breast cancer diagnostic test continues in full stride. We have expanded our oncology diagnostics team and invested EUR 1 000 000 in state-of-the-art laboratory equipment and the purchase of tissues for a large-scale retrospective clinical validation. Thanks to last year’s development and funding from the EU Horizon 2020 EIC Accelerator (Grant No. 946693), as early as 2023 we will bring more accurate diagnostics to patients with suspected breast cancer using our Multiplex8+ test.
We are happy to say that our cooperation with the Biomedical center of the Slovak Academy of Sciences is not over yet. A successful application for a research grant in Slovakia means that we are collaborating on the development of a diagnostic cellular immunity test that can be used in COVID diagnostics or for potential pandemics in the future.
About MultiplexDX
MultiplexDX is one of the most innovative biotech corporations, created to bring its revolutionary technologies to the market of personalized molecular diagnostics. The company has representation in both U.S. and European markets. The collaborators of MultiplexDX are from the world’s most prestigious scientific organizations including the National Cancer Institute, Rockefeller University, Albert Einstein University, Vanderbilt University, Cornell University, Queens University (Canada), Hebrew University of Jerusalem (Israel), and the Max Delbrück Center for Molecular Medicine (Germany).
MultiplexDX IP-based and innovative platform merges histopathology methods, biomarker quantification, visualization and gene expression with a single-cell resolution by combining MDX proprietary visual and sequencing technologies into one diagnostic test. This cross-validation approach eliminates diagnostic errors and creates 100% precise cancer profiling for each patient which allows clinicians to suggest specific, personalized cancer treatment.
Find out more about MultiplexDX on Corporate website, Facebook, LinkedIn, Twitter